- Category: Peptides
Dihexa – Peptide for neurological enhancement and repair
Share on:
What if a single peptide could rebuild the structural communication networks inside your brain – not just boost neurotransmitters temporarily, but physically generate new synaptic connections? That is the core promise of Dihexa, one of the most potent and intriguing experimental neuropeptides to emerge from academic research in recent years.
This guide covers everything you need to know: what Dihexa is, the mechanism that makes it uniquely powerful, what preclinical research suggests about its applications, how it compares to other cognitive peptides, and how Robertson Wellness and Aesthetics in Beverly Hills approaches Dihexa therapy within a medically supervised framework.
What is Dihexa?
Dihexa (chemical name: N-hexanoic-Tyr-Ile-(6) aminohexanoic amide) is a synthetic neuropeptide developed by researchers at Washington State University (WSU). It was originally designed as a potential investigational treatment for neurodegenerative conditions, particularly Alzheimer’s disease.
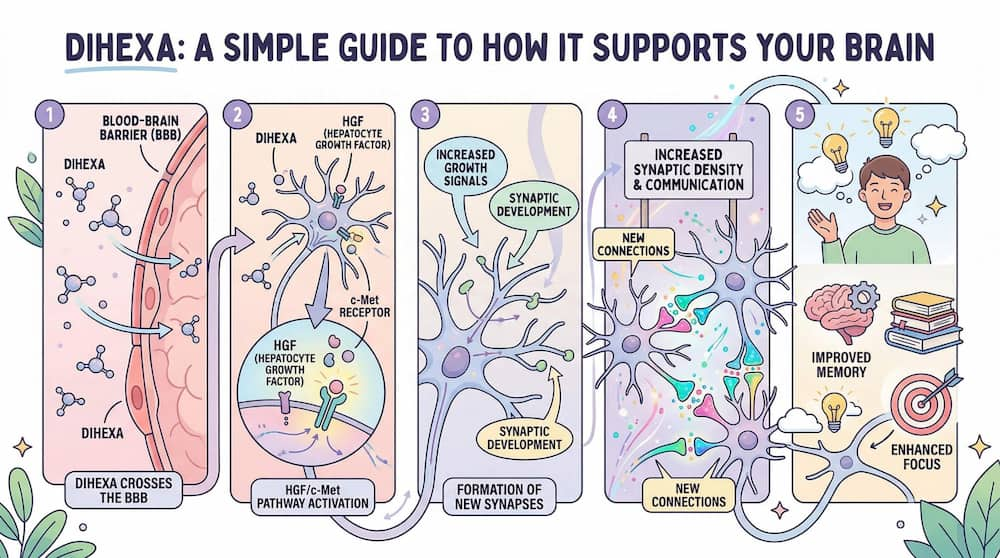
Structurally, Dihexa is derived from angiotensin IV – a naturally occurring peptide involved in brain function – but has been chemically modified to dramatically enhance its potency and its ability to penetrate the blood-brain barrier. This makes it unique among cognitive compounds: it can be taken in forms that reach the brain directly, where it interacts with specific receptor systems that drive the physical growth of neural connections.
It is important to note that Dihexa is not FDA-approved for any medical use in the United States. It remains an investigational compound, and its use in clinical settings is considered off-label. All protocols should be developed and supervised by qualified healthcare providers.
Key characteristics at a glance
| Property | Detail | Clinical significance |
|---|---|---|
| Origin | Synthetic derivative of angiotensin IV | Endogenous system – lower immune risk profile |
| Developed by | Washington State University researchers | Academic origin – well-documented research lineage |
| Primary mechanism | HGF/c-Met receptor pathway activation | Drives synaptogenesis and neuroprotection |
| Administration | Oral, topical, or subcutaneous | No injection required for most protocols |
| BBB penetration | Yes – crosses the blood-brain barrier | Direct neurological action is possible |
| FDA status | Not approved – investigational/off-label | Must be used under qualified medical supervision |
How Dihexa works – mechanism of action
Understanding what makes Dihexa different from conventional nootropics requires a basic grasp of its mechanism. Most cognitive supplements or medications work at the level of neurotransmitter modulation – they increase dopamine, acetylcholine, or serotonin availability. These effects are real but fundamentally temporary: once you stop the compound, the neurochemical balance tends to revert.
Dihexa operates at a deeper, more structural level.
The HGF/c-Met pathway
Dihexa acts as a hepatocyte growth factor (HGF) mimetic. It mimics the activity of HGF – a protein involved in cellular growth, survival, and repair – and binds to c-Met receptors present on neurons. Activating the HGF/c-Met signaling pathway initiates a cascade of events that:
-
Stimulates synaptogenesis – the physical formation of new synaptic connections between neurons
-
Promotes dendritic spine formation – the small protrusions on neurons essential for receiving signals
-
Enhances synaptic density in brain regions associated with learning and memory
-
Supports neural stem cell activity and neuroplasticity
-
Reduces neuroinflammatory signaling, offering a neuroprotective effect
Structural vs. functional enhancement
This distinction – structural vs. functional – is central to understanding Dihexa’s unique appeal. Rather than temporarily increasing cognitive performance, Dihexa may support the actual rebuilding of neural circuitry. Some researchers describe this as creating lasting changes in synaptic architecture, not just a transient neurochemical boost.
Potency compared to BDNF
In preclinical research, Dihexa has been described as potentially millions of times more potent than BDNF (brain-derived neurotrophic factor) in driving synapse growth. This is a striking finding from WSU laboratory studies, though it is crucial to note that these were preclinical results and translating such findings directly to human outcomes requires significant caution. No standardized human dosing exists, and individual responses vary considerably.
Key citation: Bhatt DL et al., Washington State University preclinical models demonstrating Dihexa’s HGF/c-Met-mediated synaptogenic activity. Patch clamp electrophysiology studies showed significantly increased synapse density in hippocampal tissue.
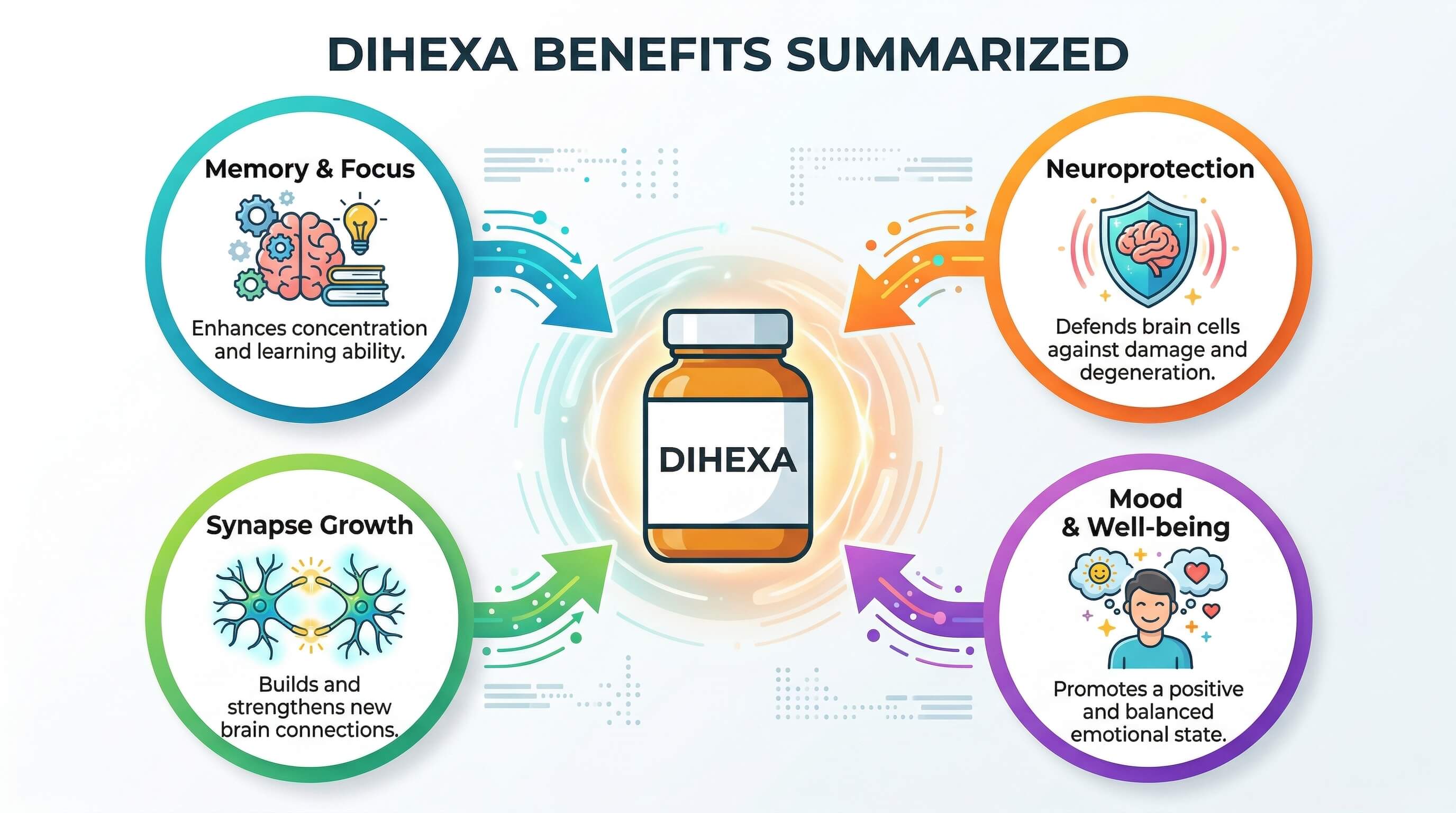
Potential benefits of Dihexa – what research suggests
Important: The following reflects findings from preclinical (animal) research and early research reports. Human clinical trial data for Dihexa remain limited. Individual results vary. These applications are investigational. Consult a qualified provider before use.
Memory formation and recall
Preclinical studies have shown Dihexa may significantly improve both short- and long-term memory in animal models with chemically induced amnesia. By increasing synaptic density and enhancing long-term potentiation (LTP) – the neurophysiological process underlying memory storage – it may strengthen the brain’s capacity to encode and retrieve information. Some individuals report improved recall clarity within 1-2 weeks, with more substantial effects developing over 4-8 weeks of use.
Neurodegeneration and Alzheimer’s disease models
One of the most compelling preclinical findings is Dihexa’s performance in Alzheimer’s disease animal models. In one widely cited study, Dihexa restored cognitive performance in a rat model of Alzheimer’s to near-normal levels – outperforming donepezil (Aricept), a standard FDA-approved Alzheimer’s treatment. Dihexa has also been patented for potential use in treating Alzheimer’s and Parkinson’s disease, though human clinical evidence remains in early stages.
Citation: Bhatt DL et al. (WSU). Dihexa reverses cognitive deficits in a scopolamine-induced Alzheimer’s model; patch-clamp studies confirm synaptogenic superiority over BDNF in hippocampal tissue.
Traumatic brain injury (TBI) support
Dihexa’s synaptogenic and neuroprotective mechanisms make it a candidate of interest for TBI recovery research. By promoting the formation of new neural connections and reducing excitotoxicity-related neuroinflammation, it may help rebuild communication pathways damaged in head injuries. This application is still primarily investigational and is explored within structured clinical protocols.
Cognitive optimization in healthy adults
Beyond therapeutic applications, a growing number of physicians and patients explore Dihexa as a proactive cognitive optimizer for healthy individuals experiencing age-related cognitive shift, brain fog, or performance demands. Its ability to enhance neuroplasticity – the brain’s capacity to reorganize itself – is the primary driver of this interest. As with all applications, this requires careful medical oversight due to the compound’s potency and the limited long-term human safety data available.
Mood and emotional well-being
Some early reports and practitioner observations suggest that Dihexa’s effects on neural connectivity may extend to emotional resilience and mood, possibly through improved prefrontal-limbic circuit function. These effects are not well-established in controlled human trials and should be considered speculative at this stage.
Dihexa vs. other cognitive peptides – a comparison
Choosing the right neuropeptide depends on your specific goals, health history, and the level of research evidence you require. Below is a comparison of Dihexa with the most commonly considered alternatives.
| Peptide | Mechanism | Admin route | Human evidence | BBB access |
|---|---|---|---|---|
| Dihexa | HGF/c-Met synaptogenesis | Oral, topical, SC | Preclinical only | Yes |
| Cerebrolysin | Multi-target neurotrophic | Injectable (IM/IV) | Extensive – 200+ trials | Yes (small peptides) |
| Semax | BDNF/ACTH analog | Nasal spray | Moderate (European) | Yes |
| Selank | GABAergic anxiolytic | Nasal spray | Moderate (Russian) | Yes |
| NSI-189 | Neurogenesis (mechanism unclear) | Oral | Limited Phase 2 data | Likely |
Cerebrolysin holds the strongest clinical research base for neurological recovery. Dihexa offers a potentially more targeted synaptogenic effect based on its unique HGF/c-Met mechanism, and its oral availability makes it more accessible than injectable peptides. Semax and Selank offer valuable complementary effects – particularly for anxiety modulation and acute cognitive clarity – with a simpler nasal spray format. Many practitioners combine these in personalized stacks.
Dihexa dosage and administration – protocol overview
Dosing guidelines are for educational reference only. No standardized dosing protocol exists for Dihexa in human clinical medicine. Always consult a qualified medical provider. Individual protocols vary significantly.
Available forms
-
Oral capsules or tablets – the most common format for wellness protocols
-
Topical cream or gel – may offer transdermal delivery
-
Powder form for compounding – allows flexible dosing by qualified pharmacies
-
Subcutaneous injection – used in some clinical settings under physician supervision
General dosage reference (investigational)
| Protocol type | Reported range | Notes |
|---|---|---|
| Cognitive optimization | 2-10 mg daily (oral) | Start low; titrate under supervision |
| Neurological recovery support | Provider-determined | Higher doses require intensive monitoring |
| Cycle structure | 4-8 weeks on / 2-4 weeks off | Cycling recommended – no indefinite use data |
| Onset of subjective effects | 1-2 weeks (some reports); 4-8 weeks full | Structural neuroplastic effects develop gradually |
Due to Dihexa’s extraordinary potency – and the theoretical concern that excessive HGF/c-Met activation could promote unwanted cellular growth in susceptible individuals – conservative dosing and careful medical supervision are strongly emphasized by experienced practitioners.
Peptyde Therapy Consultation
The recent FDA regulations have impacted the availability of certain substances essential for peptide production by compounding pharmacies. Despite these changes, we are pleased to announce that specific peptides remain available through our services, contingent upon a medical consultation.
Please book a consultation with us today.
Safety profile and important considerations
Dihexa has a limited human safety dataset. Most safety information comes from preclinical animal studies and practitioner experience reports. Based on available information, adverse effects appear mild in most users, but the lack of long-term human data means caution is warranted.
Reported side effects
-
Mild headache or fatigue – typically transient during initial use
-
Mild gastrointestinal discomfort – reported in some oral formulation users
-
Sleep disruption or irritability – potential sign of overstimulation
Theoretical safety concerns
Critical note: Because Dihexa activates the HGF/c-Met pathway – which promotes cellular growth and repair – there is a theoretical concern that it could potentially stimulate unwanted cell proliferation in individuals with active cancer or high cancer risk. While this has not been confirmed in humans, it is considered a relative contraindication by most practitioners.
-
Active malignancy: use with caution – HGF/c-Met promotes cellular growth
-
Pregnancy and breastfeeding: insufficient data; generally avoided
-
Concurrent use with other potent neuroactive compounds: manage carefully
Recommended precautions
-
Always begin under physician supervision with a full medical history review
-
Start at the lowest effective dose and titrate slowly
-
Use cycling protocols – avoid indefinite continuous use
-
Monitor for neurological overstimulation symptoms
-
Regular follow-up consultations during active protocols
Who may be a candidate for Dihexa therapy?
Candidacy for Dihexa must be determined through a comprehensive medical evaluation. General categories of individuals who explore Dihexa in supervised clinical settings include:
-
Adults experiencing age-related cognitive decline, memory concerns, or executive function challenges
-
Patients pursuing adjunct neurological support following TBI or other brain injury
-
Individuals researching evidence-informed options in the context of early neurodegenerative conditions, alongside conventional care
-
High-cognitive-demand professionals seeking evidence-informed brain optimization under medical supervision
-
Patients completing or complementing a cerebrolysin protocol who seek structural synaptogenic support
Dihexa therapy at Robertson Wellness and Aesthetics, Beverly Hills
At RWA Center in Beverly Hills, Dihexa is offered as part of a supervised neuropeptide therapy program. All peptide protocols are developed by board-certified practitioners with specialized expertise in longevity and regenerative medicine, including Biana Borchenko FNP-BC – who brings over 20 years of clinical experience and a decade of focus on peptide therapies and hormone optimization.
RWA sources its peptides from licensed U.S. compounding pharmacies meeting rigorous purity and testing standards. A full medical consultation is required before starting any peptide protocol, ensuring safety and individualization.
How Dihexa fits within RWA’s cognitive wellness framework
RWA often considers Dihexa as part of a broader neuropeptide protocol – sometimes used in sequence with Cerebrolysin (for clinical research base), Semax or Selank (for acute BDNF support and anxiety management), and NAD+ IV therapy (for cellular energy and DNA repair). The complementary mechanisms of these compounds may support a more comprehensive neurological environment than any single peptide alone.
Frequently asked questions about Dihexa
Is Dihexa FDA-approved?
No. Dihexa is not FDA-approved for any indication in the United States. It is an investigational peptide used off-label under qualified physician supervision. It has been patented for potential use in neurodegenerative conditions, but clinical drug approval processes are ongoing.
How does Dihexa work in the brain?
Dihexa activates the HGF/c-Met receptor pathway, which stimulates synaptogenesis – the formation of new synaptic connections between neurons. This structural effect on neural architecture is what distinguishes Dihexa from most nootropics, which act primarily through neurotransmitter modulation.
How long does it take for Dihexa to work?
Some individuals report improved focus and mental clarity within 1-2 weeks of starting a protocol. The structural synaptogenic effects – new neural connections – are believed to develop over 4-8 weeks of consistent use. Individual responses vary considerably based on baseline neurological status, dose, and protocol structure.
Where can I buy Dihexa?
Dihexa is available through licensed medical providers at Robertson Wellness and Aesthetics in Beverly Hills. A medical consultation is required. Visit rwacenter.com/product/dihexa/ or schedule a consultation. RWA sources exclusively from licensed U.S. compounding pharmacies. Currently shipping to California and Texas residents.
What is the price of Dihexa peptide?
Pricing depends on the protocol, formulation, and dosage determined during your consultation. Visit rwacenter.com/product/dihexa/ for current pricing, or book a consultation for a personalized assessment.
Can Dihexa be stacked with Cerebrolysin?
Some practitioners do combine Dihexa and Cerebrolysin, as their mechanisms are complementary – Cerebrolysin offers broad multi-target neurotrophic support while Dihexa targets specific synaptogenesis. However, stacking potent neuropeptides requires careful medical supervision and individualized dosing. This should not be attempted without qualified provider oversight.
Is Dihexa safe for long-term use?
Long-term human safety data for Dihexa is limited. Cycling protocols (e.g., 4-8 weeks on, followed by a rest period) are commonly recommended by practitioners. Indefinite continuous use is not advised. The theoretical concern around HGF/c-Met-mediated cellular growth means medical monitoring is particularly important.
What does Dihexa do differently than other brain peptides?
Most cognitive peptides target neurotransmitter systems or general neurotrophic support. Dihexa’s specific HGF/c-Met mechanism drives synaptogenesis at a structural level – potentially creating lasting new neural connections rather than transient neurochemical changes. Preclinical data suggest it may be orders of magnitude more potent than BDNF in this specific function, though human translation of that potency ratio is not established.
References and citations
RESEARCH CITATIONS
[1] Bhatt DL et al. Dihexa restores cognitive deficits in a rat model of Alzheimer ‘s-like impairment. Washington State University. Patch-clamp electrophysiology studies; synaptogenesis measured in hippocampal tissue.
[2] Wright JW, Harding JW. (2010). The brain angiotensin system and extracellular matrix molecules in neural plasticity, learning, and memory. Prog Neurobiol. 2010;92(1):33-45.
[3] Bhatt DL et al. The hepatocyte growth factor (HGF)/c-Met receptor system as a therapeutic target in the treatment of neurodegenerative diseases. Frontiers in Neuroscience, preliminary reports.
[4] Swolverine.com (2025). Dihexa Peptide: Benefits, Mechanism, and Cognitive Risks Explained. Peer-reviewed preclinical data synthesis.
[5] Revolution Health and Wellness (2025). Dihexa Peptide: Unlocking Brain Repair, Focus, and Memory.
Disclaimer
The information provided is for educational purposes only and is not intended as medical advice. Certain therapies discussed may be investigational and are not approved by the FDA for the diagnosis, treatment, cure, or prevention of disease. Always consult with a qualified healthcare professional before beginning any new wellness protocol.
Request a Consultation
Recommended for you
NAD therapy in Beverly Hills – IV Infusions & Cellular Health Support
If you have been researching ways to support your energy, mental clarity, and long-term cellular health, you may have come across NAD+ therapy. Once largely confined to academic research, NAD

Semax Peptide: Nasal Spray vs Injectable – Benefits and Dosage
Peptide therapy is gaining traction among health-conscious adults in the United States who are looking beyond conventional medicine for tools that may support cognitive performance, neurological resilience, and long-term brain

How Much Does NAD Cost?
If you have been researching NAD+ IV therapy, you are likely wondering whether it is worth the investment and what you should realistically expect to pay. Pricing for NAD IV

GHK-Cu: The regenerative copper peptide revolutionizing skin renewal and tissue repair
Your skin’s ability to heal, regenerate, and stay firm is driven by signals your own body produces naturally. One of the most important of those signals is GHK-Cu – a

Dihexa – Peptide for neurological enhancement and repair
What if a single peptide could rebuild the structural communication networks inside your brain – not just boost neurotransmitters temporarily, but physically generate new synaptic connections? That is the core

Cerebrolysin: The powerful neuropeptide for cognitive enhancement and brain recovery
Your brain has its own repair toolkit – but sometimes it needs support. Cerebrolysin is one of the most extensively studied neuropeptide preparations in clinical medicine, with data from more