- Category: Peptides
Cerebrolysin: The powerful neuropeptide for cognitive enhancement and brain recovery
Share on:
Your brain has its own repair toolkit – but sometimes it needs support. Cerebrolysin is one of the most extensively studied neuropeptide preparations in clinical medicine, with data from more than 200 clinical trials and approximately 15,000 patients across multiple neurological conditions. Whether you are exploring options for post-stroke rehabilitation, researching evidence-based cognitive support, or simply trying to understand what Cerebrolysin is and whether it may be right for you, this guide covers everything.
Below, you will find a plain-English breakdown of what Cerebrolysin is, how it works, what the research suggests, how it compares to other neuropeptides, and how Robertson Wellness and Aesthetics in Beverly Hills approaches Cerebrolysin peptide therapy.
What is Cerebrolysin?
Cerebrolysin is a lipid-free neuropeptide preparation created through standardized enzymatic breakdown of purified porcine (pig) brain proteins. The result is a complex mixture of low-molecular-weight neuropeptides and free amino acids – all below 10,000 Daltons in size. That small molecular size is the defining advantage: these peptide fragments can cross the blood-brain barrier and interact directly with neurons, something that most endogenous neurotrophic factors cannot do on their own.
Unlike single-compound nootropics that target one neurotransmitter pathway, Cerebrolysin delivers a cocktail of bioactive peptides that mimics the effects of several endogenous growth factors simultaneously.
What does Cerebrolysin contain?
The peptide complex contains fragments associated with the following neurotrophic factors:
-
BDNF (brain-derived neurotrophic factor) – supports neuronal survival, synaptic regulation, learning, and memory formation
-
NGF (nerve growth factor) – promotes survival of sensory neurons and supports acetylcholine signaling
-
GDNF (glial cell line-derived neurotrophic factor) – aids axonal regeneration and helps prevent neuronal apoptosis (programmed cell death)
-
CNTF (ciliary neurotrophic factor) – plays a role in protecting motor neurons from degeneration
-
P21 – a neuropeptide fragment associated with enhanced neurogenesis and brain tissue adaptability
-
Enkephalins – endogenous opioid peptides involved in stress and pain regulation
-
Orexin – influences arousal, sleep-wake cycles, appetite, and cognitive motivation
A brief history of Cerebrolysin
Cerebrolysin was first developed in 1949 by Austrian professor Gerhart Harrer and received regulatory approval in Austria in 1954. Over the following decades, it became widely used in Europe and Asia. Today it is approved in more than 45 countries for conditions including ischemic stroke, traumatic brain injury (TBI), and various forms of dementia.
In the United States, Cerebrolysin is not currently approved by the FDA for any medical indication. It may be used off-label by qualified medical providers as an investigational therapy, and availability through compounding pharmacies is subject to evolving regulatory guidance.
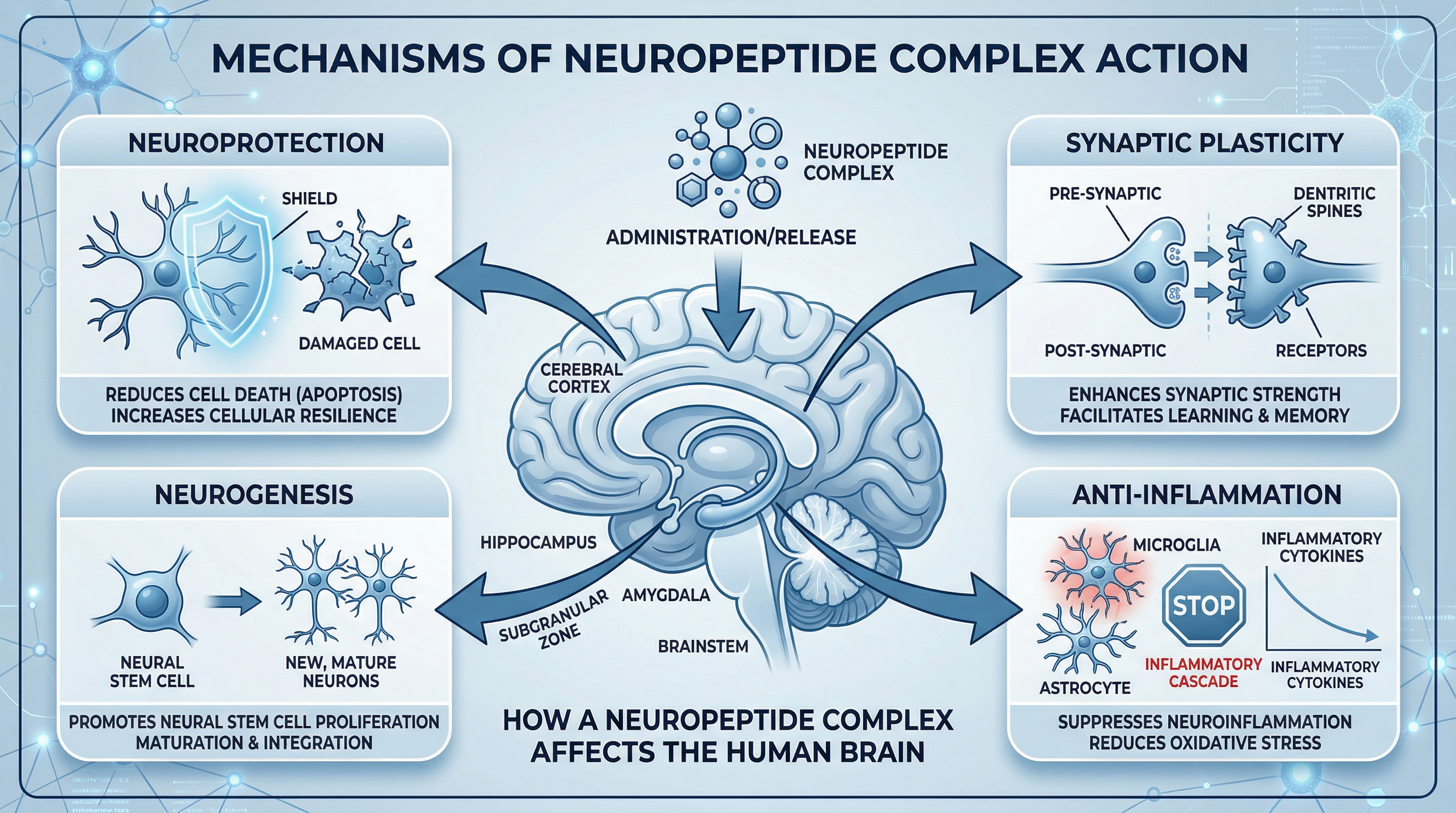
How Cerebrolysin works – mechanisms of action
Cerebrolysin is described as a multimodal neuropeptide, meaning it acts through several pathways simultaneously rather than a single mechanism. This is one of its most clinically significant features, and one reason researchers continue to study it for complex neurological conditions where single-target interventions have shown limited efficacy.
Supporting neurotrophic signaling
The peptide fragments in Cerebrolysin actively stimulate the production of the brain’s own endogenous neurotrophic factors while simultaneously providing direct neurotrophic support. This dual action – external peptide supply plus stimulation of endogenous upregulation – may produce more sustained neurological effects than supplementing a single growth factor alone.
Neuroprotection from injury cascades
After a neurological injury – such as stroke, TBI, or the chronic damage seen in neurodegeneration – a cascade of secondary injury events unfolds: excitotoxicity, uncontrolled apoptosis, oxidative stress, and neuroinflammation. Research suggests that Cerebrolysin may help interrupt this cascade through inhibition of apoptosis, modulation of synaptic plasticity, and regulation of inflammatory responses. It may also reduce the formation of toxic protein aggregates, which play a role in neurodegeneration.
Neurogenesis and synaptic density
Studies indicate Cerebrolysin may promote the formation of new neurons (neurogenesis) and new synaptic connections – particularly in the hippocampus, the brain region central to memory and learning. Preclinical research has shown it may significantly increase the number of new synapses in the hippocampus and promote neurovascular protection and repair.
It has also been shown in research to activate the CREB/PGC-1-alpha pathway, which plays a role in suppressing neuroinflammation by reducing pro-inflammatory cytokines and promoting anti-inflammatory mediators.
Supporting the brain’s self-repair signaling
Cerebrolysin has been shown to modify two major signaling systems: the neurotrophic factor (NTF) pathway and the sonic hedgehog (Shh) signaling pathway. These pathways regulate neurogenesis, angiogenesis (new blood vessel formation), myelination, dendrite development, axonal sprouting, and the integrity of the neurovascular unit – the critical connection between brain cells and blood supply.
Peptyde Therapy Consultation
The recent FDA regulations have impacted the availability of certain substances essential for peptide production by compounding pharmacies. Despite these changes, we are pleased to announce that specific peptides remain available through our services, contingent upon a medical consultation.
Please book a consultation with us today.
Potential benefits of Cerebrolysin – what the research suggests
Important: Individual results vary. Many applications of Cerebrolysin in the United States are considered investigational or off-label. The following reflects published research findings – not guaranteed outcomes. Consult a qualified provider for personalized guidance.
Cognitive function and mental clarity
Clinical trials have examined Cerebrolysin’s potential role in cognitive performance across multiple patient populations. Some patients with mild cognitive impairment, age-related memory decline, and early-stage dementia have demonstrated improvements in memory, attention, and daily functioning in controlled studies. Many individuals report experiencing improved mental clarity, better focus, and reduced brain fog – though these are subjective outcomes and individual results vary significantly.
Post-stroke recovery support
Cerebrolysin has been studied in multiple randomized controlled trials in ischemic stroke patients. The CARS trial – a significant multicenter randomized controlled trial – found that patients receiving Cerebrolysin early in stroke rehabilitation experienced improved functional outcomes and global recovery compared to those who did not. Researchers noted potential improvements in motor function and measures of independence.
It is important to note that evidence in this specific area is mixed. A 2023 Cochrane review found that Cerebrolysin provided no clear benefit for preventing death in acute ischemic stroke, and noted a potentially higher rate of spontaneous adverse events in some trial subgroups. This context underscores the importance of medical supervision and individualized evaluation.
Traumatic brain injury (TBI) rehabilitation
A 2025 review in ScienceDirect examining 10 published clinical studies in more than 1,900 TBI patients found that Cerebrolysin may support motor skills, cognitive abilities, and overall functional outcomes when used as part of a structured rehabilitation protocol. Preclinical studies have shown it may help decrease infarct volume, reduce brain edema, and preserve neuronal integrity in TBI models. These findings are encouraging, though more large-scale trials are needed.
Neurodegeneration – Alzheimer’s disease and dementia
A review of 15 randomized controlled trials including over 2,400 subjects found that higher doses of Cerebrolysin were associated with reduced psychological symptoms and potentially slower progression in patients with Alzheimer’s disease and dementia. Research also suggests possible cognitive benefits in vascular dementia. These applications are actively being studied – Cerebrolysin is not a cure for Alzheimer’s disease, and these findings represent early-stage or supporting evidence.
Proactive neuroprotection and long-term brain health
Beyond acute neurological events, some clinicians explore Cerebrolysin as part of a proactive longevity and brain health protocol. Its role in upregulating BDNF – sometimes described as the brain’s key growth factor for neuronal health – and its capacity to support neuroplasticity are primary reasons practitioners consider it for healthy adults seeking cognitive optimization. The research base for this specific application remains more limited than therapeutic uses.
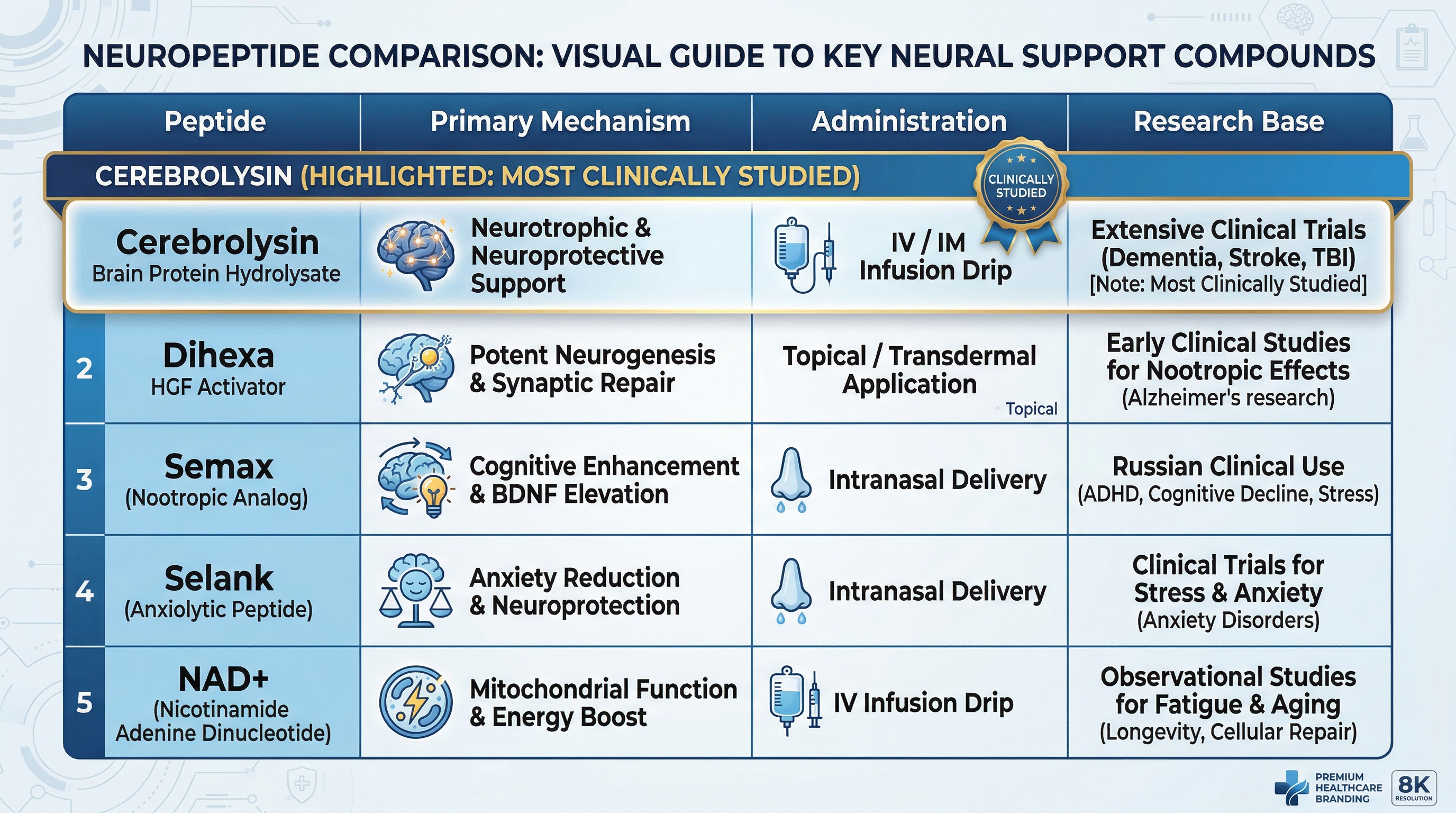
Cerebrolysin vs. other neuropeptides – how does it compare?
Understanding Cerebrolysin in context helps set realistic expectations and identify whether it may work well as a standalone therapy or as part of a broader neuropeptide protocol.
| Peptide | Primary mechanism | Administration | Research base |
|---|---|---|---|
| Cerebrolysin | Multi-target neurotrophic support | Injectable (IM or IV) | Extensive (200+ trials, 15,000+ patients) |
| Dihexa | Neurogenesis via HGF/MET pathway | Oral or topical | Limited human data |
| Semax | BDNF upregulation, ACTH-derived | Nasal spray | Moderate (primarily European/Russian research) |
| Selank | Anxiolytic, GABA modulation | Nasal spray | Moderate |
| NAD+ IV | Cellular energy, sirtuins, and DNA repair | IV infusion | Growing (complementary to neuropeptides) |
Cerebrolysin has the deepest clinical research base of any neuropeptide in this category. Dihexa may offer potentially more potent neurogenic effects but has significantly less human trial data. Semax and Selank are excellent complementary options for cognitive support and mood regulation, with different administration methods and price points. NAD+ IV therapy supports a different but complementary set of mechanisms – primarily cellular energy and DNA repair – and is often used alongside neuropeptide protocols.
Cerebrolysin dosage and administration – what protocols look like
The following information is provided for educational purposes only. Dosage and treatment protocols should always be determined by a qualified healthcare provider based on your individual health history and goals.
How Cerebrolysin is administered
Cerebrolysin is administered exclusively by injection – either intramuscular (IM) for lower doses or intravenous (IV) infusion for higher therapeutic doses. Oral administration is not effective because the peptide fragments are broken down in the digestive tract before they can reach systemic circulation.
Typical dosage guidance
| Protocol type | Typical dosage | Cycle length (general guidance) |
|---|---|---|
| Cognitive optimization/wellness | 5-10 mL (IM injection) | 5-20 sessions, 1-3 cycles per year |
| Stroke / acute neurological recovery | 10-50 mL (slow IV infusion over 15 min) | Intensive, under specialist supervision |
| Neurodegeneration support | Provider-determined based on assessment | Ongoing – periodic re-evaluation required |
Standard IV infusion protocol: Doses between 10 mL and 50 mL are diluted in 100 mL of standard infusion solution (0.9% sodium chloride, Ringer’s solution, or 5% glucose) and administered as a slow IV infusion over approximately 15 minutes. Cerebrolysin should not be mixed with other medications in the same infusion.
IM injection: Doses up to 5 mL can be administered undiluted as a slow intramuscular injection over approximately 3 minutes.
Treatment cycle structure
Standard protocols typically involve cycles of 5-20 daily or alternating-day sessions followed by a rest period. For cognitive optimization and wellness protocols, many practitioners suggest 1-3 cycles per year. Neurological recovery protocols may require more intensive and extended regimens under close specialist supervision. Because Cerebrolysin’s effects involve neuroplastic changes that develop over time, some patients report that benefits continue to consolidate after the active treatment period ends.
Safety profile and potential side effects
Cerebrolysin has a well-documented safety record based on decades of clinical use and extensive post-marketing surveillance. The European Medicines Agency (EMA) classifies it in the safe category. Adverse effects reported in clinical trials are generally mild and transient.
Reported side effects
-
Mild local irritation or discomfort at the injection site
-
Headache (typically mild and short-lived)
-
Nausea or dizziness in some patients
-
Sweating
-
Mild fever – more commonly reported in post-stroke use, generally manageable with standard antipyretics
Contraindications
Cerebrolysin is not recommended for individuals with:
-
Active epilepsy or seizure disorders
-
Severe renal (kidney) impairment
-
Known hypersensitivity to any component of the formulation
Note: A 2023 Cochrane review noted a potentially higher rate of spontaneous adverse events in some stroke trial subgroups. As with all investigational therapies, full disclosure of your medical history to your provider is essential before starting any Cerebrolysin protocol.
Who may be a candidate for Cerebrolysin therapy?
Cerebrolysin is currently used off-label in the United States for a range of applications. Individual candidacy always requires a thorough medical evaluation. The following are general categories of individuals who explore Cerebrolysin therapy in clinical settings:
-
Adults experiencing age-related cognitive decline, mild memory concerns, or subjective brain fog
-
Patients in post-stroke or post-TBI rehabilitation seeking additional neurological support alongside standard rehabilitation
-
Individuals with early-stage neurodegenerative conditions, explored in coordination with existing standard-of-care approaches
-
High-performing professionals, executives, or biohackers seeking evidence-based cognitive optimization
-
Patients pursuing proactive brain health and longevity protocols with medical supervision
Cerebrolysin peptide therapy at Robertson Wellness and Aesthetics, Beverly Hills
At Robertson Wellness and Aesthetics in Beverly Hills, Cerebrolysin is offered as part of a fully personalized peptide therapy program. All treatments are provided under the supervision of board-certified practitioners with deep expertise in longevity medicine, peptide therapy, and neurological wellness.
Biana Borchenko, FNP-BC – a board-certified family nurse practitioner with over 20 years of clinical experience including a decade specializing in peptide therapies, hormone optimization, and longevity medicine – leads many of RWA’s advanced peptide protocols. As a member of the American Academy of Anti-Aging Medicine (A4M), Biana brings functional and regenerative medicine expertise to each patient consultation.
What to expect at your consultation
Every Cerebrolysin protocol begins with a comprehensive medical consultation. Your provider will review your complete health history, current medications, existing conditions, and personal wellness goals to determine whether Cerebrolysin is appropriate and how to structure an individualized protocol. Dosing, cycle length, and complementary therapies are customized to each patient.
Cerebrolysin may be administered at the RWA clinic via intramuscular injection or IV infusion, depending on your protocol. Certain patients may be provided with take-home options following initial supervised sessions, subject to provider assessment.
Complementary neuropeptide and IV therapies at RWA
RWA offers a range of neuropeptide and IV therapies that may complement or be combined with Cerebrolysin as part of a comprehensive brain health approach:
Many patients explore Cerebrolysin as part of a broader cognitive wellness or longevity stack. Combining Cerebrolysin with NAD+ infusions, complementary neuropeptides, and lifestyle protocols – including regular aerobic exercise (a potent natural BDNF booster), quality sleep optimization, and anti-inflammatory nutrition – may support a more comprehensive environment for brain health optimization.
Book your consultation
Robertson Wellness and Aesthetics is located at 8920 Wilshire Blvd, Suite 320, Beverly Hills, CA 90211. Our team is available to discuss whether Cerebrolysin or any other peptide therapy may be appropriate for your individual health goals.
Frequently asked questions about Cerebrolysin
Is Cerebrolysin FDA–approved?
No. As of 2026, Cerebrolysin is not FDA-approved for any indication in the United States. It is approved in more than 45 countries, including Austria, Germany, Russia, China, and South Korea for conditions including stroke, traumatic brain injury, and dementia. In the US, it may be used off-label by qualified medical providers as an investigational therapy. Always consult a licensed healthcare provider before use.
How is Cerebrolysin administered?
Cerebrolysin is given exclusively by injection – either intramuscular (IM) for smaller doses or intravenous (IV) infusion for larger therapeutic doses. It is not effective when taken orally because the peptide fragments are broken down in the digestive tract before reaching systemic circulation.
How long does it take to notice effects?
Individual results vary significantly. Some patients in clinical studies reported improvements within the first treatment cycle (5-20 sessions). Others required multiple cycles. Cognitive and functional effects may continue to develop after the treatment period ends, as neuroplastic changes consolidate over time. Some benefits may be subtle, and formal cognitive assessments can be helpful in tracking progress.
Can Cerebrolysin be stacked with other peptides?
Some practitioners combine Cerebrolysin with other neuropeptides such as Semax, Selank, or Dihexa as part of a comprehensive brain health protocol. Certain clinicians also pair it with NAD+ IV therapy for broader cellular and neurological support. Stacking decisions should always be made under medical supervision, as interactions and individual tolerability vary.
What does Cerebrolysin cost at RWA?
Pricing depends on the specific protocol, dosage, and number of sessions required. Visit rwacenter.com/product/Cerebrolysin/ or schedule a consultation at RWA for a personalized cost assessment.
Is Cerebrolysin safe for long-term use?
Cerebrolysin has a documented safety record from decades of clinical application in Europe and Asia. The EMA classifies it as safe, and adverse effects reported in clinical trials are generally mild and transient. However, independent long-term self-administration without medical oversight is not recommended. Contraindications include epilepsy, severe renal impairment, and known hypersensitivity to the compound.
How does Cerebrolysin compare to other nootropics?
Cerebrolysin has a significantly deeper clinical research base than most cognitive supplements or nootropics – with data from 200+ clinical trials and approximately 15,000 patients. Unlike single-target supplements, its multimodal mechanism addresses multiple aspects of brain health simultaneously: neuroprotection, neurotrophic support, anti-neuroinflammation, and neuroplasticity. This makes it distinct from racetams, lion’s mane, citicoline, and most other commonly available brain supplements.
Can Cerebrolysin help with brain fog?
Some patients report reduced brain fog and improved mental clarity during Cerebrolysin treatment cycles. These reports are consistent with its studied mechanisms – including reduced neuroinflammation and BDNF upregulation. However, brain fog has many causes, and individual results vary. A medical consultation to identify underlying contributors to cognitive symptoms is always the recommended first step.
Is Cerebrolysin a peptide?
Technically, Cerebrolysin is a peptide complex rather than a single peptide. It contains a mixture of low-molecular-weight neuropeptides and free amino acids derived from porcine brain proteins. The active fragments include components that mimic BDNF, NGF, GDNF, CNTF, and other neurotrophic factors. This complex composition is what distinguishes Cerebrolysin from simpler synthetic peptides.
Where can I buy Cerebrolysin?
Cerebrolysin is available through licensed medical providers in the United States. Robertson Wellness and Aesthetics offers Cerebrolysin peptide through its peptide therapy program (subject to medical consultation and availability to California and Texas residents). Visit rwacenter.com/product/Cerebrolysin/ to learn more or to book a consultation.
Disclaimer
The information provided is for educational purposes only and is not intended as medical advice. Certain therapies discussed may be investigational and are not approved by the FDA for the diagnosis, treatment, cure, or prevention of disease. Always consult with a qualified healthcare professional before beginning any new wellness protocol.
Request a Consultation
Recommended for you

NAD+ Dosage Guide: How Much Should You Take by IV, Injection, and More
“How much NAD+ should I take?” is one of the most common questions patients bring to a consultation – and there is no single correct answer. NAD+ dosage depends on
Migraine IV Treatment: The Cocktail Approach to Acute Migraine Relief
When a severe migraine hits, oral medications often fall short. The reason is part of the migraine itself: during an active attack, the stomach slows down in a process called

NAD+ Injections: Benefits, Types, and What to Expect
NAD+ injections have become one of the most flexible ways to support cellular health outside of a clinical IV setting. They deliver a faster session, can be continued at home
Beauty IV drip therapy: What’s Inside And How It Supports Skin, Hair, And Nail Wellness
When people think about taking care of their skin, hair, and nails, they usually think about topical products, salon treatments, or oral supplements. Beauty IV drip therapy offers a different
Myers Cocktail IV Therapy: Complete Guide to Ingredients, Benefits, and What to Expect
First developed in the 1970s by Dr. John Myers, a Baltimore-based physician, the Myers cocktail has become one of the most recognizable formulations in intravenous nutrient therapy. Today, it is

Hangover IV Therapy: a Science-Informed Look at Fast Recovery and Hydration Support
A long evening out, an important celebration, a weekend that ran longer than planned – the next morning often arrives with a pounding head, dry mouth, fatigue, and a sensitive